What Drives Uniformity In The Electroplating Process And How To Control It
Fundamentals of Uniformity in the Electroplating Process
The electroplating process stands as a key part of surface engineering. It affects how metals handle mechanical, chemical, and environmental pressures. Even coating thickness matters more than looks. It sets durability, conductivity, and corrosion resistance. In factory settings, you cannot split uniformity from electrochemical control. Both grow side by side as settings change during plating. I recall a time when a small shift in current led to big differences in part quality—it’s a reminder that balance is everything here.
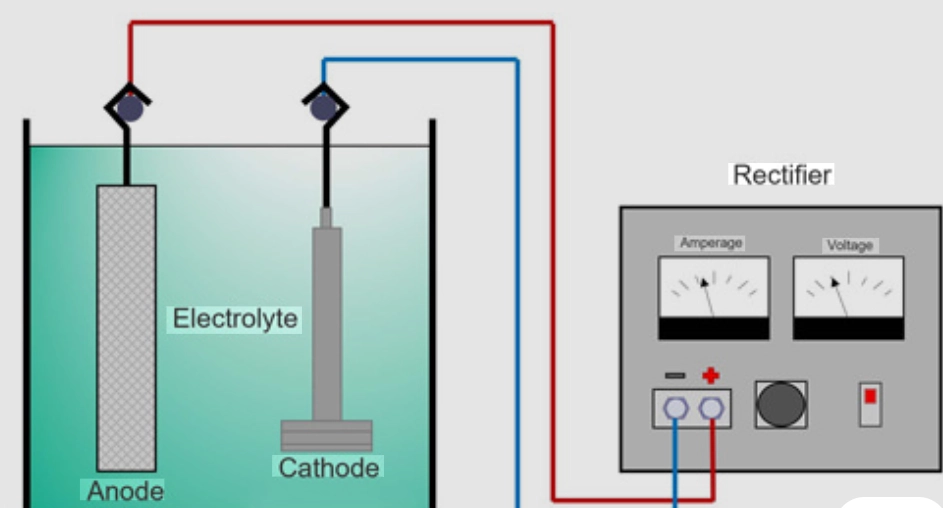
The Concept of Coating Uniformity
Plating uniformity means how smoothly a metal layer builds over a base material’s surface. At first glance, a plated item might look even and shiny. But close-up checks often show tiny differences in thickness. The link between layer depth, current density, and shape is straightforward. Spots near the anode or with pointy edges pull in more current density. This results in thicker layers there. On the other hand, sunken spots get less current. So, they end up with thinner coatings. Such uneven spread can hurt corrosion resistance. Thin areas rust quicker. It also changes wear strength or electrical flow where exactness counts a lot. For instance, in car parts, this might mean faster wear on edges after just a few months of use.
Electrochemical Principles Governing Uniform Deposition
Steady deposition depends on steady electrochemical actions in the plating tank. Ions travel through the liquid to reach the cathode surface. That travel decides where metal ions land. The voltage spread in the tank shapes where reduction reactions happen most. Cathodic polarization and overpotential matter a great deal. When overpotential rises past a limit, ion reduction speeds up in a curved way. This causes buildup in certain spots. Throwing power measures how well a mix plates into dips. It shows this effect clearly. Good throwing power brings better evenness on tricky shapes. Current efficiency tells how much power goes straight to metal buildup. The rest might fuel side reactions, like hydrogen release.
Factors Influencing Uniformity During Electroplating
Uniformity does not come about on its own. Shape, mix, warmth, and flow in the tank all shape it. Each one ties into the others. Shift one thing, and it often touches several results later on. Think of it like a chain reaction in a busy workshop— one tweak ripples through the whole line.
Current Density Distribution Across Complex Geometries
In everyday plating setups, items seldom have basic forms. Corners, openings, threads—they all bend electric field paths. This makes areas with focused current density. Edges often build up too much. That’s because electrons take the quickest routes through the conducting liquid. Tools like finite element analysis map these spreads. They predict uneven spots before making starts. You can fix part position or rack setup to even things out a lot. In one factory I heard about, rotating racks by 15 degrees cut thickness gaps by half.
Solution Chemistry and Its Role in Deposit Consistency
The liquid’s makeup sets how ions shift and act at the surface. Metal ion levels need to stay steady. If they drop near busy current spots, you get rough or scorched layers. Buffer setups hold pH in tight ranges. Local hydrogen making can sour small areas at the cathode edge. Additives like brighteners fine-tune grain setup. They help even starting points across the surface. Levelers hold back growth at high spots. Suppressors slow action in strong current zones. Together, they make smoother ends. Without them, deposits might look dull, like in older plating jobs before additives became standard.
Temperature Control in the Plating Bath
Warmth touches nearly all parts of the process. It sways ion speed, spread rate, reaction pace, and even crystal direction. If one end of the tank gets warmer than the other—from bad flow or heater spots—you see thickness changes on items plated at once. Handling warmth differences keeps steady form in groups. Bath stirring often mixes heat too. It holds both mix and warmth even. A steady 40 degrees Celsius, for example, can make a big difference in daily output consistency.
Agitation and Mass Transport Mechanisms
Stirring keeps ions ready at the cathode face. It does this where they count most. Methods include hand stirring, air bubbles, or pump flows. Good movement stops buildups from forming near edge layers. A heavy edge layer slows spread. It leads to flat, dull areas. Too much stirring might shake fragile items. Or it could leave swirl marks. Finding the right strength fits the item’s shape. Small dips need soft flow. Flat open areas handle stronger currents. In practice, gentle air sparging works wonders for intricate jewelry pieces without scratching them.
Methods to Control and Enhance Coating Uniformity
After you understand what causes uneven buildup, ways to manage it get simpler. They start with holder design and go to power wave choices.
Design Considerations for Fixtures and Racking Systems
Holders place parts so current routes balance around each shape. Bad setup makes hidden spots with little plating. Extra anodes near dip features spread current better. Shields by edges cut extra plating on bumps. Even clean links between rack parts and hooks stop local voltage falls. Those falls twist current paths. Simple fixes like this can save hours of rework in a production run.
Optimization of Electrical Parameters
Current Type Selection: DC vs Pulse Plating
Standard direct current (DC) plating gives constant push. But it fights tricky shapes. Ions run low faster near strong field spots. Pulse plating switches on and off—or even backward pulses. This renews ion levels at the edge during breaks. The method sharpens grain size. It boosts cover in openings or grooves. You control starting actions with wave changes.
Voltage Regulation Strategies
Choosing constant voltage or constant current fits the aims. Constant voltage is easy. Yet it risks uneven spread if link resistance shifts. Constant current keeps steady output. Still, it needs close watch on voltage drops along wires and joins. This avoids hot points that speed local buildup. In high-volume lines, sticking to constant current often proves more reliable over long shifts.
Analytical Techniques for Monitoring Uniformity
Thickness Measurement Methods
You cannot fix what you do not check right. X-ray fluorescence (XRF) mapping gives touch-free depth outlines across faces. It hits micrometer accuracy. Coulometric stripping offers straight electrochemical checks. It melts known coating amounts. Magnetic induction suits iron-based bases under nickel or copper layers best.
Surface Morphology Analysis
Scanning electron microscopy (SEM) shows grain edges and hole changes hidden from eye checks. Atomic force microscopy (AFM) gauges tiny roughness. That ties close to work traits like rub or stick strength. These tools have caught issues early, preventing batch rejects worth thousands in one case I know of.
Common Sources of Coating Nonuniformity and Their Mitigation
Well-planned setups still face changes. These come from shape effects to dirt problems. You must tackle them step by step, not just when they pop up. It’s like routine maintenance—better to stay ahead.
Localized High Current Density Areas
Pointy corners pull electric fields like magnets. Bumps plate quicker while dips fall back. Shields from non-reactive stuff guide field lines more evenly. Shaped anodes close to item outlines cut gaps further. They shorten ion routes the same across faces. Using these, a shop once evened out plating on threaded bolts that were tricky before.
Contaminants in the Plating Bath
Bits from additives wearing out can mess with crystal patterns. Metal bits spark bad reactions. This leads to bumps or holes. Regular filtering with carbon clears organic layers. Now-and-then dummy plating pulls stray metal ions. It stops clear flaws. Skipping this can turn a smooth bath into a nightmare fast.
Inconsistent Pre-treatment Processes
Surface ready work lays the base for all that comes next. Any leftover from cleaning fats or rust blocks stick in spots. This causes bare areas down the line. Set steps with base cleaners then acid starts give steady wet habits. So each group begins with the same surface power. Uniform pre-treatment has saved many a run from failure.
Advanced Control Strategies for Industrial Electroplating Operations
Today’s making spots lean on auto tools. They boost speed and steadiness that hand work cannot match now. Automation feels like a game-changer, especially in 24-hour plants.
Integration of Process Automation and Real-Time Monitoring
Sensors watch bath mix all the time. They measure metal ion counts, warmth spreads, pH shifts. Data goes to control setups. These tweak add pumps or heaters when things stray. Live watching holds quality over work times. It cuts out worker guesses that once caused run differences. With sensors, variations drop to under 5% in most setups.
Modeling and Simulation Tools for Predictive Control
Finite element analysis (FEA) pictures electric field strength before tool making. This saves money on test fixes later in real setups. Learning machines trained on batch data spot links between setting wiggles and depth maps. They allow ahead tuning over after-the-fact fixes when flaws show. Simulations have predicted issues in gear plating, avoiding weeks of downtime.
Sustainable Practices in Maintaining Uniform Electroplating Quality
Reusing liquids via built-in renew units holds makeup steady. It slashes chemical throwaway way below old dump schedules from years back. Keeping tight loops on power in versus layer out lets plants hit green rules and close layer limits at once. That’s a mix once seen as impossible. Plus, it cuts costs—recycling can save up to 30% on supplies yearly.
FAQ
Q1: What causes uneven coating thickness during electroplating?
A: Uneven coating usually results from irregular current density distribution caused by complex geometry or poor racking alignment within the bath.
Q2: How does temperature affect plating uniformity?
A: Temperature changes influence ion mobility and reaction rates; inconsistent heating leads to varied deposit morphology across different zones in the tank.
Q3: Why are additives important in maintaining uniform coatings?
A: Additives modify local electrochemical behavior—brighteners promote fine grains while levelers suppress buildup at high-current points improving overall smoothness.
Q4: What tools help analyze plating uniformity effectively?
A: XRF mapping provides precise thickness data while SEM imaging exposes microstructural differences critical for diagnosing uneven deposits.
Q5: How do automated systems enhance process stability?
A: Automation uses sensors for real-time feedback adjusting variables dynamically ensuring consistent coating quality without manual intervention errors.

