What Makes The Hot Dip Galvanizing Process Critical For Long-Term Durability
Fundamentals of the Hot Dip Galvanizing Process
Hot dip galvanizing is a basic metal process. It puts a strong zinc layer on steel to stop rust. This way has improved over more than 100 years. It mixes chemical changes, exact heat control, and smart planning. The result is a coating that lasts many years in tough spots. For builders and coating experts, learning these steps is key. It helps get steady results and make steel items last longer.
Overview of the Metallurgical Bonding Mechanism
The hot dip galvanizing process uses a metal bond, not just a stuck-on layer. Steel goes into hot zinc, and slow spread reactions happen at the join. This makes a set of mixed metal layers between iron and zinc. These layers are Gamma (Γ), Delta (δ), Zeta (ζ), and Eta (η). They form a tight link right into the steel. The coating works as a wall against harm. It also gives extra guard through cathodic protection. So, if you scratch the zinc, the nearby zinc rusts first. This keeps the steel safe from rust. Think about a fence in salty air; the zinc takes the hit instead.
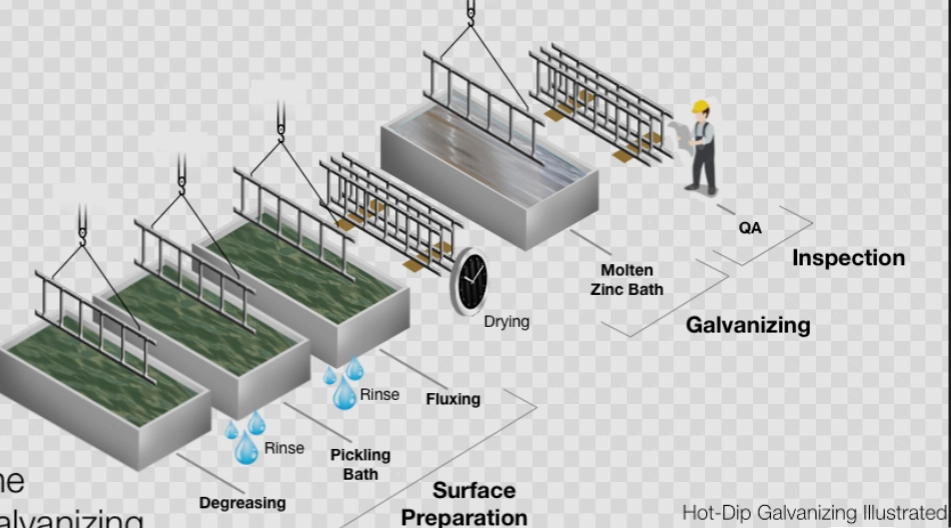
Key Stages in the Hot Dip Galvanizing Sequence
The order of steps sets the coating’s strength. Main parts are cleaning the surface, adding flux, dipping in hot zinc, and letting it cool. Each part affects how well it sticks, how thick it is, and how even it looks. The bath heat stays near 450°C. Dip time gets watched closely. This keeps the reactions steady and the surface smooth. But even tiny changes can make the coating bumpy or too thick in spots. In a real shop, workers check these things all the time to avoid problems.
Surface Preparation: The Foundation of Coating Quality
Steel must be spotless before it hits the hot zinc. Things like grease, rust, or factory dirt block the bond. This prep step often decides if the coating works well or breaks down fast. I’ve seen jobs fail just because of a little overlooked dirt.
Importance of Proper Cleaning and Degreasing
Cleaning starts with a basic soap wash to get rid of fats and dirt bits. Then acid soak removes rust and hard scales. Leftover junk can leave blank spots in the zinc layer. Those spots let rust in quick. In factories, they watch the clean tanks for strength and warmth. Wrong settings might leave thin films that cause sticking issues later. For example, if the acid is too weak, scale stays and you end up with weak spots on a bridge beam.
Fluxing and Its Role in Surface Reactivity
After the wash, flux stops air from rusting the steel again before the dip. Flux often has zinc ammonium chloride. It helps the hot zinc spread well on the surface. Drying the flux right is vital too. Wet spots can make the zinc splash wild when you dip. Skilled workers look for an even salt cover to know it’s ready. Miss this, and you get messy results, like on those big tank parts they do in coastal plants.
Immersion in Molten Zinc: Core Reaction Phase
This part turns plain steel into rust-proof stuff through steady metal reactions. The whole hot dip galvanizing process hinges on keeping the zinc bath just right in mix and heat. Workers adjust it daily based on what comes in.
Bath Composition and Temperature Control
The zinc bath holds steady at about 450°C. It’s mostly pure zinc. Sometimes they add a bit of aluminum or nickel to make it look better or cut down on waste bits. But stuff like lead can mess up the surface, making it rough. Even heat across the bath means the spread happens the same on all shapes. That’s big for handling large builds, like tower frames. In one case, a shop fixed uneven heat and saved a whole batch from redo.
Formation of Intermetallic Layers During Dipping
Gamma (Γ), Delta (δ), Zeta (ζ), and Eta (η) Layers Explained
Steel meets hot zinc and builds alloy layers one by one, starting from the base out. First is Gamma (Fe₃Zn₁₀), then Delta (FeZn₇), Zeta (FeZn₁₃), and Eta (pure Zn). Each has its own toughness and bend level. Inside ones are tough but can snap. Outside ones bend easy and stay soft. How fast they grow ties to silicon in the steel, called the Sandelin effect. Dip length and bath mix matter too. Pulling out at the right speed stops drips and keeps it smooth. On curved pipes, this makes a big difference in the finish.
Post-Galvanizing Inspection and Quality Control
After cooling, checks make sure parts match rules like ASTM A123 or ISO 1461. Quality watch keeps things the same batch to batch. This is must for big builds where lasting counts, such as road supports that face weather year-round.
Visual Examination and Thickness Measurement Techniques
Eye checks spot even cover, shine match, drips, or bare parts. Tools like magnetic readers or weight checks measure how thick it is on different areas. They stack up against base needs for the steel type and weather class. If it’s too thin from dirt or quick dip, they might redo it or fix with zinc paint. In practice, a 5-micron shortfall on a pole can mean trouble after a few rains.
Adhesion Testing and Coating Performance Evaluation
Stick tests see if the bond holds under bends or hits. Quick rust tests, like salt mist, mimic years of wear in short time. Results help plan upkeep for things like bridges, poles, pipes, or towers in salty air. One test showed a coated beam holding up 20% better than painted ones after 500 hours of spray.
Design Considerations for Effective Galvanizing Application
Smart planning eases the galvanizing and boosts outcomes. Builders think about shape, air holes, weld strength, and steel makeup way before making. It’s like prepping a recipe; skip it, and the dish flops.
Structural Design Factors Influencing Coating Quality
Air vents let gases out during dip. Without them, bubbles trap and leave gaps in hollow spots. Drain holes let zinc flow out full after pull-out. This cuts big build-ups at bends or tips. Round sharp corners a touch. They cool fast and get thin layers. For a real example, a vented guardrail section coated even, while a sealed one had hidden weak points.
Material Selection and Compatibility with Galvanizing Process Parameters
Influence of Steel Chemistry on Coating Behavior
What’s in the steel shapes how it acts with hot zinc. Silicon from 0.03% to 0.12% speeds things up a lot. That’s the Sandelin effect. It makes thick, dull layers instead of shiny ones. High phosphorus does close, leading to breakable mixes that crack easy. So, check the makeup first. Then tweak heat or dip time for best output. Shops often test samples to dodge this, saving time and cash.
Long-Term Durability Achieved Through Hot Dip Galvanizing
These zinc coatings beat most others. They mix guard ways that team up for years, even by the sea or in factory smoke. It’s reliable, but remember, no coating lasts forever without some check-ups now and then.
Synergistic Protection Mechanisms Against Corrosion
The layer blocks air and wet from the steel. Plus, it self-fixes at cuts through cathodic work. Zinc goes first, saving the base. This pair setup is why power line towers stay good for 50 years outside, no big fixes needed. In wet spots, it holds where paint peels off quick.
Performance Comparison with Alternative Protective Systems
Evaluating Cost-to-Lifecycle Efficiency
Paint needs fresh coats every 5 to 10 years. Thin plate coatings don’t cover much. Hot dip galvanizing wins on long-run cost. Real stats show less fix money over time. It fights wear, wet shifts, salt from roads, and sun rays well, like on highways. Life checks prove it’s green too, using few resources over the years. Take a 30-year bridge job; galvanizing cuts total spend by 40% versus paint touch-ups.
FAQ
Q1: What is the typical temperature used during hot dip galvanizing?
A: The molten zinc bath is maintained around 450°C to promote proper diffusion between iron and zinc while avoiding excessive alloy growth.
Q2: Why does silicon content affect coating thickness?
A: Silicon accelerates reaction kinetics between steel and molten zinc causing thicker intermetallic layers known as the Sandelin effect.
Q3: How can poor surface preparation impact final coating quality?
A: Residual oils or oxides prevent metallurgical bonding leading to bare patches that quickly corrode once exposed outdoors.
Q4: What standards govern inspection for galvanized coatings?
A: Common references include ASTM A123 for structural products and ISO 1461 which specify visual criteria plus minimum thickness values.
Q5: How long can galvanized steel last without maintenance?
A: Depending on environment severity it can perform effectively for 30–70 years thanks to combined barrier and cathodic protection mechanisms inherent in its coating structure.

